The Philippine Genome Center is pleased to announce the appointment of Dr. Windell L. Rivera as its new Executive Director. Dr. Rivera is a Professor of Microbiology at the Institute of Biology, College of Science, and UP Scientist III at the University of the Philippines Diliman. He also serves as an Academician at the National […]

Decoding 1,001 Philippine traditional rice varieties (TRVs)
The joint initiative stands to deliver a huge amount of data from the genome sequences of the 1001 Philippine TRVs. This will be made available to the public via the PH Rice GDB. From strengthening local expertise in crop genomics and bioinformatics, to providing more research opportunities—the program anticipates its potential in increasing and strengthening research output and capacity. The 1001 Rice Genome (1k1RG) Program is a promise to the future of rice in the Philippines as it ultimately aims to benefit farmers through accelerating the improvement and production of rice varieties.

Clinical Geneticist-Molecular Genetic Pathologist now heads the Philippine Genome Center
The Philippine Genome Center of the UP System welcomes Felicitas L. Lacbawan, MD who used to teach at the Department of Human Biochemistry and Molecular Biology at the UP College of Medicine. Dr. Lacbawan, a former chief medical officer for genetics and genomics in the US rejoined the University to rise to the challenge of […]

Dr. Cynthia Saloma of PGC is a recipient of the Presidential Lingkod Bayan award
The PGC team’s dedication and resilience are a testament to the importance of scientific research and its ability to overcome even the most difficult of circumstances. #PGCParaSaBayan

Register now—Prevention and Action: Developing Tools to Protect Children from Exploitation
Aimed at creating awareness on the new developments in forensic investigation in the fight against sexual abuse and exploitation, the webinar “Prevention and Action: Developing Tools to Protect Children from Exploitation” is happening on March 22,2023 at 12:30 pm-5:00 pm.
Medical doctors in the Child Protection Unit, social welfare workers, law enforcement personnel, forensic researchers, and the general community are invited and may register at https://tinyurl.com/FGRP3-Webinar

Register to the PBEF-IB webinar on April 29, 2022
A webinar hosted by the Program on Biodiversity, Ethnicity, and Forensics of the Philippine Genome Center and the Institute of Biology, University of the Philippines Diliman Focusing on Philippine Biodiversity: The Philippine BioGenome Project The Philippine BioGenome Project (PBGP) is a research initiative headed by the PGC Program on Biodiversity, Ethnicity, and Forensics (PBEF). The […]

Methods used in microbial forensics and epidemiological investigations for stronger health systems
Establishing and maintaining a national microbial forensic system to strengthen capacities in conducting forensic and epidemiological investigations should be prioritised by all countries, accompanied by a national policy that sets the legislative framework and provides for the system’s financial requirements.

Challenges on the Development of Biodiversity Biobanks: The Living Archives of Biodiversity
In this review, we highlight the necessity for biodiversity biorepositories by presenting their significant role in health, biodiversity, linking of big data, other translational research, and biodiversity conservation efforts.
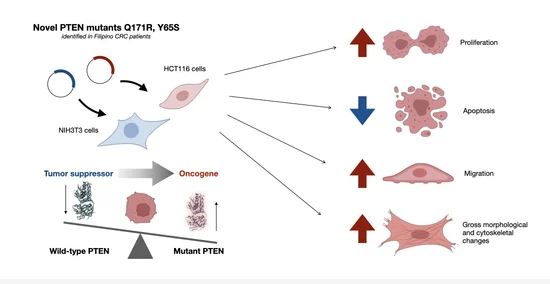
The Novel Phosphatase Domain Mutations Q171R and Y65S Switch PTEN from Tumor Suppressor to Oncogene
Phosphatase and tensin homolog deleted on chromosome 10, or PTEN, is a well-characterized tumor suppressor with both lipid and protein phosphatase activities. PTEN is often downregulated by epigenetic mechanisms such as hypermethylation, which leads to constitutive activation of the PI3K–Akt pathway.

High rates of tenofovir failure in a CRF01_AE-predominant HIV epidemic in the Philippines
The Philippines has the fastest growing HIV epidemic in the Asia-Pacific. This increase was accompanied by a shift in the predominant HIV subtype from B to CRF01_AE. Increasing evidence points to a difference in treatment responses between subtypes. We examined treatment failure and acquired drug resistance (ADR) in people living with HIV (PLHIVs) after one year on antiretrovirals (ARVs).

