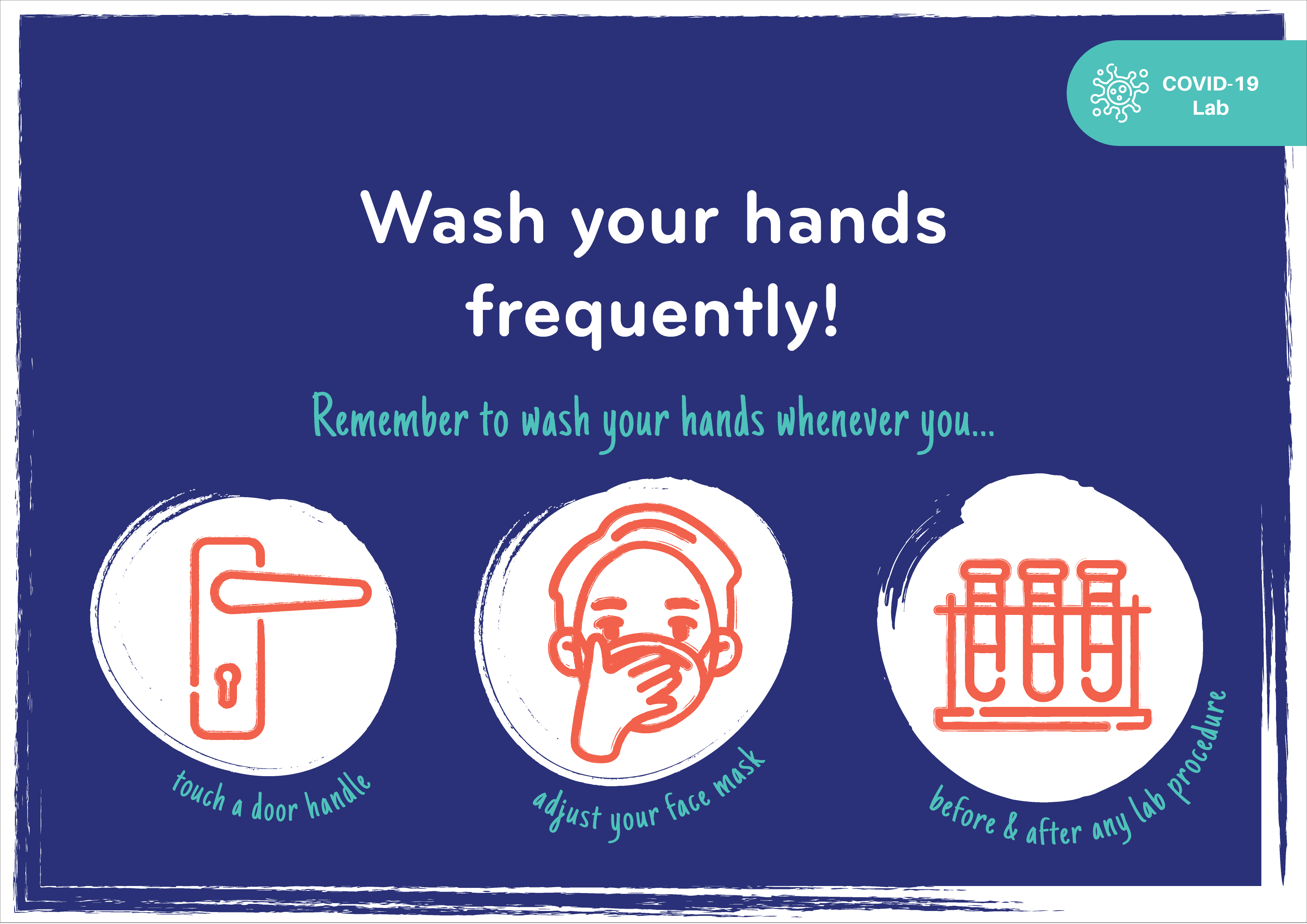
PGC shares its COVID-19 laboratory safety tips and precautions through the illustrations below.

PGC Core Facility for Bioinformatics releases six (6) genome sequences of SARS-CoV-2 from Philippine samples between March 22-28, 2020
The Philippine Genome Center (PGC) thru its Core Facility for Bioinformatics (CFB) releases today to the global community through the GISAID database six (6) genomes of the SARS-CoV-2 sequences from Philippine samples taken in Metro Manila between 26 to 28 March 2020.

PGC’s Clinical Genomics Laboratory certified to conduct testing for COVID-19 (SARS-CoV-2) by Real-Time PCR
Upon completing and passing the Research Institute for Tropical Medicine’s (RITM) proficiency testing for SARS-CoV-2 detection by Real-Time PCR, the Clinical Genomics Laboratory (CGL) of the Philippine Genome Center is now certified to perform independent testing for COVID-19. The certification is released as of April 23, 2020 by the RITM-DOH.

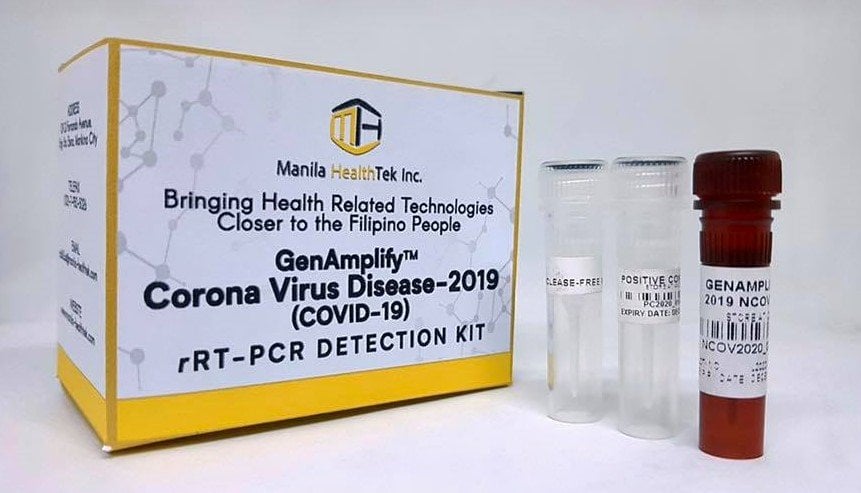
Steps in using the locally made rRT-PCR Detection Kit
How does COVID-19 Testing work? See the general checklist and step-by-step procedure on how the COVID-19 testing works, using the Philippine-made GenAmplify test kit.

Salin sa Filipinong mga terminolohiya kaugnay ng COVID-19: COVID-19, Pathogen, Virus Coronavirus
Halaw sa salin ni Prof. Eag Narvaez, UPD-FPP

COVID-19 UPDATES
PGC’s preparation and response in addressing the COVID-19 national health emergency Press Release: Philippine Genome Center gears up to verify presence of 2019-nCoV through whole genome sequencing 01/30/2020 The Philippine Genome Center is ready to verify the presence of 2019-nCoV through whole genome sequencing. With the use of Next Generation DNA/RNA Sequencing equipment (NGS) available […]
The Philippine Genome Center: Stockpiling for COVID-19
According to PGC Executive Director Cynthia Palmes-Saloma, the kit was made possible after genetic experts from others countries used Next Generation DNA Sequencing in trying to understand the nature of the COVID-19 virus. Among the facilities of the center is its DNA Sequencing Laboratory which was established in 2013.

Genome Center to help detect nCoV via whole-genome sequencing
The Centers for Disease Control and Prevention (CDC) describes whole-genome sequencing as a “laboratory procedure that determines the order of bases in the genome of an organism in one process.”
According to CDC, scientists conduct data analysis, which is the fourth step of whole-genome sequencing, to compare “bacterial sequences and identify differences.”